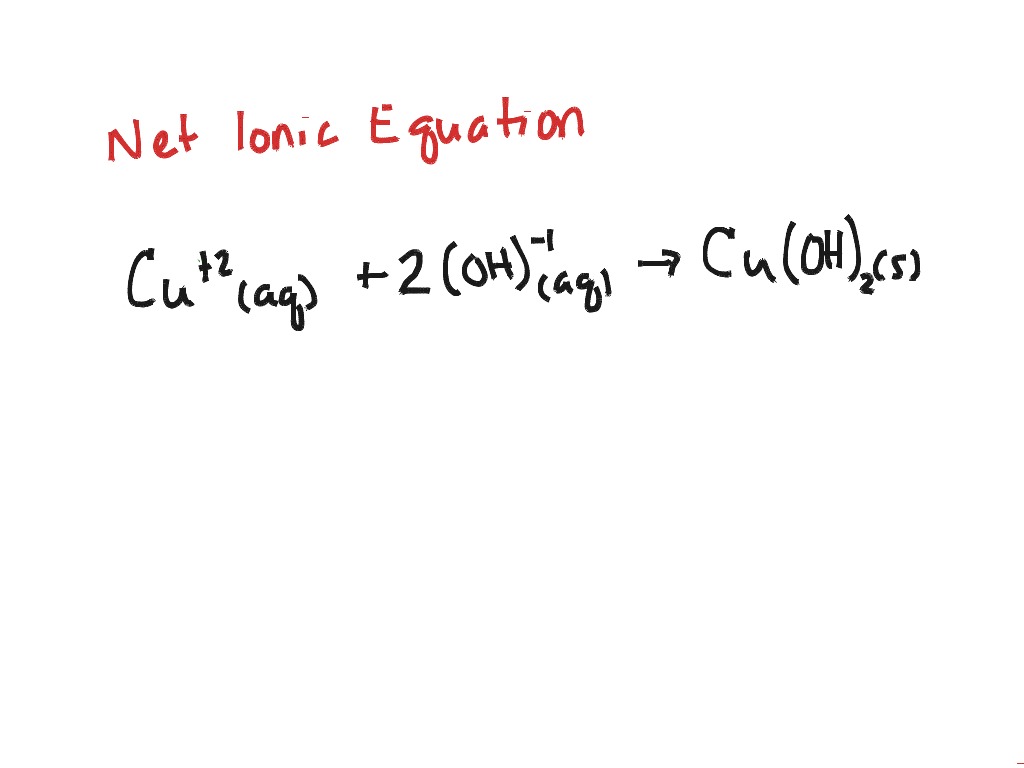
Acid and base strengths are determined by their abilities to dissociate (ions breaking apart when dissolved).Common bases that you may have dealt with include: Ammonia, sodium bicarbonate, sodium hydroxide, magnesium hydroxide, calcium hydroxide, calcium carbonate (coffee is acidic… you’ll notice this when you drink bad coffee).Basic solutions are often called “alkaline solutions.”.Bases are commonly known as substances that are corrosive, have a slippery feel, taste bitter, turn litmus paper blue, and react with acids to form salts.Ex: NaOH + H+ Na+ + H2O NH3 + H+ NH4+ Mg(OH)2 + 2H+ Mg+ + 2H2O NaHCO3 + H+ Na+ + H2CO3 There are no naming rules for bases It is often called a “proton acceptor.” Notice that these two definitions are very different. a substance that can accept a hydrogen ion (H+) from a proton donor.Common acids that you deal with every day include: Acetic acid, carbonic acid, citric acid, maleic acid, fumaric acid, ascorbic acid, acetylsalicylic acid, amino acids, hydrochloric acid.Acids are well known as substances that are corrosive, sour tasting, turn litmus paper a red color, and react with bases and metals to form salts.Ex: HBrO3 – bromic acid HBrO2 – bromous acid If it ends in –ite, then the acid is named by the oxyanion and the suffix of –ous.
If it ends in –ate, then the acid is named by the oxyanion and the suffix of –ic. Name these by noting the name of the oxyanion and following it with the word acid.Oxyacids – acids of polyatomic ions that contain oxygen.Ex: HCl – hydrochloric acid Usually binary means 2, but exceptions are made when it is an acid of a polyatomic ion that does not contain oxygen Ex: HCN – hydrocyanic acid Name these by using hydro- for the hydrogen and the name of the second element with ending of –ic, then following it up with the word, acid.Binary acids – usually acids that are made of hydrogen and one other substance.Hydronium ions are created when an acid is dissolved into water.It is another way to understand how hydrogen ions, H+, move around in a solution.It is often called a “proton donor.” Notice the slight difference in wording, but essentially they mean the same thing… Ex: HCl H+ + Cl- HNO3 H+ + NO3- HCH3CO2 H+ + CH3CO2. An acid is a substance that can transfer a hydrogen ion (H+), also called a proton, or Hydronium (H3O+) to another substance.The ions that are not involved in the reaction are called spectator ions and are removed from the reaction.Net ionic equations are used to show only the chemicals and ions involved in a chemical reaction in order to simplify information about a reaction.
